Stearyl Methacrylate: Application Characteristics & Polymerization Research
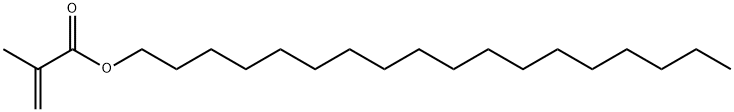
Stearyl Methacrylate is an ester of Methacrylic Acid used as a raw material component in synthesizing polymers. In order to prevent polymerization, Stearyl Methacrylate must always be stored under air, and never under inert gases. The presence of oxygen is required for the stabilizer to function effectively. It has to contain a stabilizer and the storage temperature must not exceed 35 °C. Under these conditions, a storage stability of one year can be expected upon delivery. In order to minimize the likelihood of overstorage, the storage procedure should strictly follow the “first-in-first-out” principle. For extended storage periods over 4 weeks it is advisable to replenish the dissolved oxygen content.

Effect of Stearyl Methacrylate Comonomer on Dental Resins
Tooth hard tissue loss due to dental caries is a common problem in oral health. According to the World Health Organization (WHO), the vast majority of children and all adults worldwide have tooth decay. Teeth play a significant role in human life, both physically and psychologically. Dental composites have many superior advantages, but there are also many challenges to overcome. Dental composite restorations need to be replaced over time for different reasons. These include marginal defects, secondary caries, fractures and discoloration of the restoration material. Stearyl methacrylate (SMA), which has high hydrophobicity due to its long alkyl chain, was used as a hydrophobic monomer in the study, which has not been tried in dental resins before. SMA also contains double bonds that can react with double bonds of Bis-GMA and TEGDMA. SMA is a mono-functional monomer. Mono-functional monomers lead to the formation of more linear polymers than multifunctional monomers, reducing the amount of crosslinking. Therefore, the addition of a mono-functional monomer causes a decrease in the mechanical properties of the polymer structure obtained. However, secondary interactions arising from the long alkyl chain in the monomers added to the composition are expected to have a positive effect on the mechanical properties. It is thought that the mechanical improvement due to the long alkyl chain of stearyl methacrylate can tolerate the deterioration in mechanical properties caused by mono-functionality.[1]
SMA is a monofunctional monomer. According to the literature, the presence of monofunctional monomers in bifunctional monomers increases the DC. In this study, contrary to the literature, DC values decreased with stearyl methacrylate addition. In our formulation, SMA was added instead of TEGDMA. So, as the amount of SMA increased, the amount of TEGDMA decreased, which resulted in a relatively increased amount of Bis-GMA. This caused a decrease in the DC values. This is because TEGDMA, with low viscosity, enhances the reactivity of monomers due to the increase in mobility of monomers when it is added to Bis-GMA. The addition of stearyl methacrylate up to 25 wt% to the TEGDMA and Bis-GMA mixture reduced the WS value of the polymers. However, mechanical properties reduced with the increasing SMA ratio due to the monofunctional structure of SMA. According to the results, the amount of SMA is critical for the best composition. For high mechanical properties, the SMA amount has to be lower than 25 wt%, and at the same time, it has to be higher than 12.5 wt% for lower water sorption. It is seen that the addition of SMA slightly increases the thermal stability of the polymer obtained. stearyl methacrylate changes the thermal degradation profile of the polymer.
Graft Polymerization of Stearyl Methacrylate on PET Track-Etched Membranes
Water has been one of the most indispensable commodities used by all kinds of industries. During the manufacturing, separation and purification processes in petrochemical, pharmaceutical, metallurgical, oil, and gas industries large volumes of oily wastewater are being generated. Graft polymerization can be implemented by using the methods of thermo-, radiation-, plasma- and photo-initiation. One of the solutions to this problem is to increase the pore diameter of membranes and develop methods for hydrophobing membranes with large pore diameters. On one hand, stearyl methacrylate (SM) is a long side chain monomer with hydrophobic properties that can be used to improve the water-repellent properties of materials. On the other hand, there are currently no articles of SM being grafted to PET TeMs. At the same time, its accessibility, low price and high hydrophobic properties (up to 149°) make it an excellent monomer for imparting stable hydrophobic properties to PET TeMs with large pore diameters up to 3 µm. In this article, we have applied the method of photo-induced graft polymerization of stearyl methacrylate (SM) on PET TeMs. Hydrophobized PET TeMs with different pore sizes were tested in the separation of oil-water emulsions using the model systems hexadecane–water and chloroform–water.[2]
In this study, we presented the results of PET TeMs hydrophobization by photo-induced graft polymerization of stearyl methacrylate. The effect of monomer concentration and time of UV irradiation was studied, and optimal conditions leading to the highest water contact angle with minimal change in pore structure were found. PET TeMs with pore diameters ranging from 350 nm (pore density of 1 × 108) to 3.05 µm (pore density of 1 × 106) were tested in oil–water emulsion separation by using hexadecane–water and chloroform–water model systems as emulsions. Then membranes were placed in a solution of stearyl methacrylate in 2-propanol with concentration range of 1–45% and irradiated under the UV-lamp OSRAM Ultra Vitalux E27. Membranes have shown stable fluxes and separation degrees during eight filtration cycles for emulsion separation. Membranes with large pore diameters showed a maximum flux value of 1100 mL/m2∙s for chloroform–water emulsion at a vacuum pressure of 900 mbar and 573 mL/m2∙s for hexadecane–water emulsion at a vacuum pressure of 600 mbar. The increase in fluxes for hydrophobic membranes with a pore size of 3.05 μm and a pore density of 1 × 106 was almost three times greater than for membranes with a pore size of 350 nm and a pore density of 1 × 108.
References
[1]Karadag M, Dolekcekic E, Erdem M, Özcan M. Effect of Stearyl Methacrylate Comonomer on the Mechanical and Physical Properties of Dimethacrylate-Based Dental Resins. Materials (Basel). 2024 Aug 21;17(16):4136. doi: 10.3390/ma17164136. PMID: 39203314; PMCID: PMC11356059.
[2]Yeszhanov AB, Muslimova IB, Melnikova GB, Petrovskaya AS, Seitbayev AS, Chizhik SA, Zhappar NK, Korolkov IV, Güven O, Zdorovets MV. Graft Polymerization of Stearyl Methacrylate on PET Track-Etched Membranes for Oil-Water Separation. Polymers (Basel). 2022 Jul 26;14(15):3015. doi: 10.3390/polym14153015. PMID: 35893980; PMCID: PMC9331679.
You may like
See also
Lastest Price from Stearyl Methacrylate manufacturers

US $6.00/kg2025-04-21
- CAS:
- 32360-05-7
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 2000KG/Month

US $30.00-10.00/KG2025-04-15
- CAS:
- 32360-05-7
- Min. Order:
- 50KG
- Purity:
- 99%
- Supply Ability:
- 500000kg